
It is used to determine the efficiencies of systems like heat-engines and heat-pumps. The Rankine cycle deals with heat being added at a constant pressure leading to isentropic expansion and heat extraction at a constant pressure leading to isentropic compression. He developed the complete theory of the steam engine and placed a huge emphasis on practical applications of his work. It was named after William John Macquorn Rankine, a Scottish engineer and physicist shown below: The Rankine cycle is the ideal model used to describe the operation of steam heat engines. Each of these iso-lines represents the thermodynamic quantity at a certain constant value.
Ternary diagram equilibrium lines series#
Additional thermodymic quantities may each be illustrated in increments as a series of lines - curved, straight, or a combination of curved and straight. In a two-dimensional graph, two of the thermodynamic quantities may be shown on the horizontal and vertical axes. s) for water/steam or for a refrigerant are commonly used to illustrate thermodynamic cycles such as a Carnot cycle, Rankine cycle, or vapor-compression refrigeration cycle. For example, single-component graphs of Temperature vs. Examples of such thermodynamic properties include specific volume, specific enthalpy, or specific entropy. In addition to just temperature or pressure, other thermodynamic properties may be graphed in phase diagrams. This reflects the fact that ice has a lower density than water, which is an unusual property for a material. In some parts of the phase diagram for water the solid-liquid phase boundary has a negative slope (especially the portion corresponding to standard pressure). This is due to the solid phase having a higher density than the liquid, so that increasing the pressure increases the melting point the temperature at which a substance melts. Noteworthy is that the solid-liquid phase boundary in the phase diagram of most substances has a positive slope. However, the solid-liquid phase boundary can only end in a critical point this way if the solid and liquid phases have the same symmetry group. Thus, the liquid and gaseous phases can blend continuously into each other. When going from the liquid to the gaseous phase, one usually crosses the phase boundary, but it is possible to choose a path that never crosses the boundary by going to the right of the critical point. The existence of the liquid-gas critical point reveals a slight ambiguity in the above definitions.
This reflects the fact that, at extremely high temperatures and pressures, the liquid and gaseous phases become indistinguishable, in what is known as a supercritical fluid. Instead, it terminates at a point on the phase diagram called the critical point. In the phase diagram, the phase boundary between liquid and gas does not continue indefinitely. The phases are separated by lines of non-analyticity, where phase transitions occur, which are called phase boundaries.
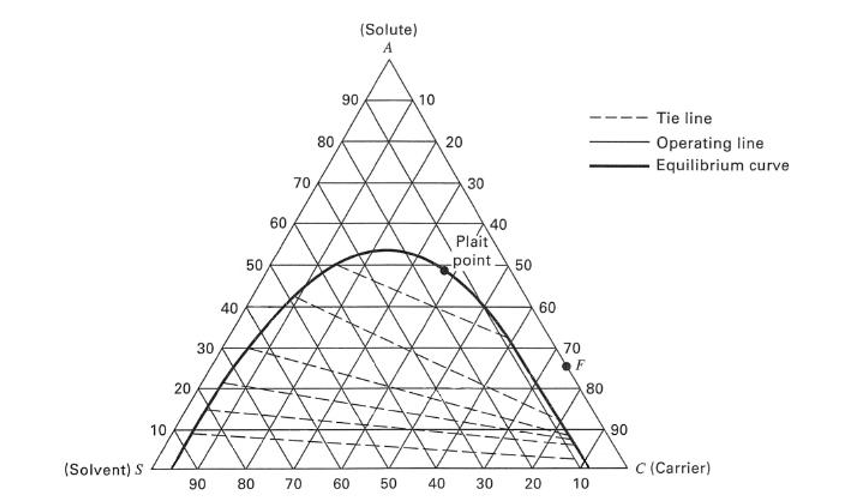
Ternary diagram equilibrium lines free#
The open spaces, where the free energy is analytic, correspond to the phases. The green lines mark the freezing point and the blue line the boiling point, showing how they vary with pressure.The markings on the phase diagram show the points where the free energy is non-analytic. The dotted line gives the anomalous behaviour of water. The picture shown above is a typical phase diagram. The phase diagram shows, in pressure-temperature space, the lines of equilibrium or phase boundaries between the three phases of solid, liquid, and gas. The axes correspond to the pressure and temperature. A phase diagramThe simplest phase diagrams are pressure-temperature diagrams of a single simple substance, such as water.
